KYMRIAH, AN ANTI-CD19 CAR-T THERAPY
INTRODUCTION
Kymriah (Novartis) is an advanced therapy medicine that belongs to the gene therapy
product category. It is approved for B-cell acute lymphoblastic leukemia (ALL) in children and
young adults under the age of 25 whose cancer did not respond to previous treatment, has
relapsed two or more times or has relapsed after stem cell transplantation².
ALL is a type of cancer of the blood and bone marrow and the most common cancer in children. The term «acute» is derived from the fact that the disease progresses rapidly and creates immature
aberrant lymphocytes³. Kymriah is also approved to treat adults with Diffuse large B-cell lymphoma (DLBCL) whose cancer has recurred or did not respond after two or more prior treatments ². DLBCL is a type of non-Hodgkin lymphoma (NHL), a cancer of the lymphatic system characterized by abnormal proliferation of B lymphocytes⁴ . As both diseases are rare, Kymriah was recognized as an orphan medicine (a medicine used to treat rare diseases) for B-cell ALL and DLBCL in 2014 and 2016 respectively².
DRUG CHARACTERISTICS
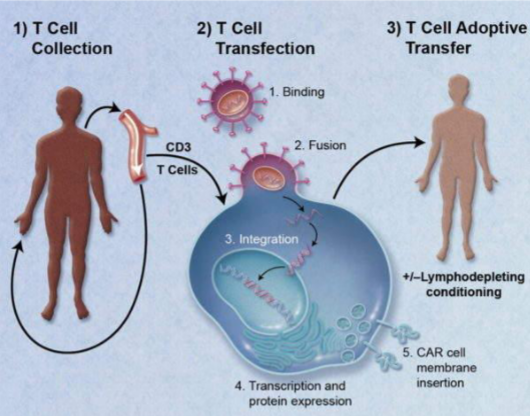
Kymriah is made from the patient’s own T cells, which are extracted from the blood and genetically modified in the laboratory to produce a Chimeric Antigen Receptor (CAR). This CAR is specifically designed to bind to CD19 protein², expressed by most B cells. This molecule is required for the survival, differentiation, and activation of B cells in the peripheral immune system⁵. The modified T cells are injected into a vein as a single infusion, following short-term chemotherapy to deplete the patient’s white blood cells. Immediately before infusion, they are administered paracetamol and antihistamines to reduce the risk of infusion reactions ².
APPROVAL
Kymriah’s pivotal study in B-cell ALL involved 92 children and young adults (ages 3–25) whose cancer had recurred or did not respond after previous treatment. About 66% of patients showed a complete remission within three months after treatment. This was better than what was seen with other drugs, such as clofarabine, blinatumomab, or a combination of clofarabine, cyclophosphamide, and etoposide. After 12 months of treatment, the probability of survival was 70%². Patients with B-cell ALL have a poor prognosis (5-year overall survival between 56 and 66%⁶). As improvements with Kymriah were better than with other medicines for this condition, it received a marketing authorization valid throughout Europe and the US for ALL in 2017².
RISK ASSESSMENT
Most patients experience serious side effects, which may include cytokine release
syndrome. It is a potentially life-threatening illness that can cause fever, vomiting, shortness of
breath, pain, and low blood pressure. It may also cause a decrease in platelets, hemoglobin,
neutrophils, and lymphocytes. However, these conditions can be managed with appropriate
measures such as close monitoring for ten days after treatment and staying close to a specialist
clinic for at least four weeks after treatment. Also, in the case of cytokine release syndrome,
another drug (tocilizumab) and emergency equipment can be used².

Need more information? Talk with us
Looking for project funding? Talk with us
REFERENCES
2018;
KYMRIAH, AN ANTI-CD19 CAR-T THERAPY
KYMRIAH, AN ANTI-CD19 CAR-T THERAPY
Share This Post
More To Explore

Uncategorized
Tecartus
INTRODUCTION Tecartus is a new advanced therapy approved to treat adults with mantle cell lymphoma (MCL)¹. This disease is a lymphatic system cancer. Specifically, it
Diego Fajardo
mayo 10, 2022

Uncategorized
KYMRIAH, AN ANTI-CD19 CAR-T THERAPY
INTRODUCTION Kymriah (Novartis) is an advanced therapy medicine that belongs to the gene therapyproduct category. It is approved for B-cell acute lymphoblastic leukemia (ALL) in
Diego Fajardo
abril 19, 2022


